Let’s talk about what everyone is asking about — but few are fully understanding.
Ozempic. Wegovy. Mounjaro. These medications are everywhere right now — in the news, in conversations with GPs, and in the social feeds of millions of people looking for answers about their weight and health.
They belong to a class of drugs called GLP-1 receptor agonists, and I want to be clear from the outset: they are genuinely effective. Clinical trials show average body weight reductions of 15% or more. For people with type 2 diabetes or significant obesity, that is a meaningful result. I’m not here to tell you these drugs are dangerous or that you should avoid them.
But I am a naturopath, nutritionist, and metabolic health practitioner — and I’ve spent a lot of time researching these medications. What I keep seeing is a gap between what the science shows and what patients are actually being told.
Most people going on these medications — and many of the clinicians prescribing them — are missing information that changes everything about how they should be used.
This post is my attempt to bridge that gap. We’ll cover how GLP-1 drugs actually work, the risks that rarely make the headlines, the biology of fat loss that almost nobody talks about, and what I consider to be a much smarter way to use these medications when they’re genuinely needed.
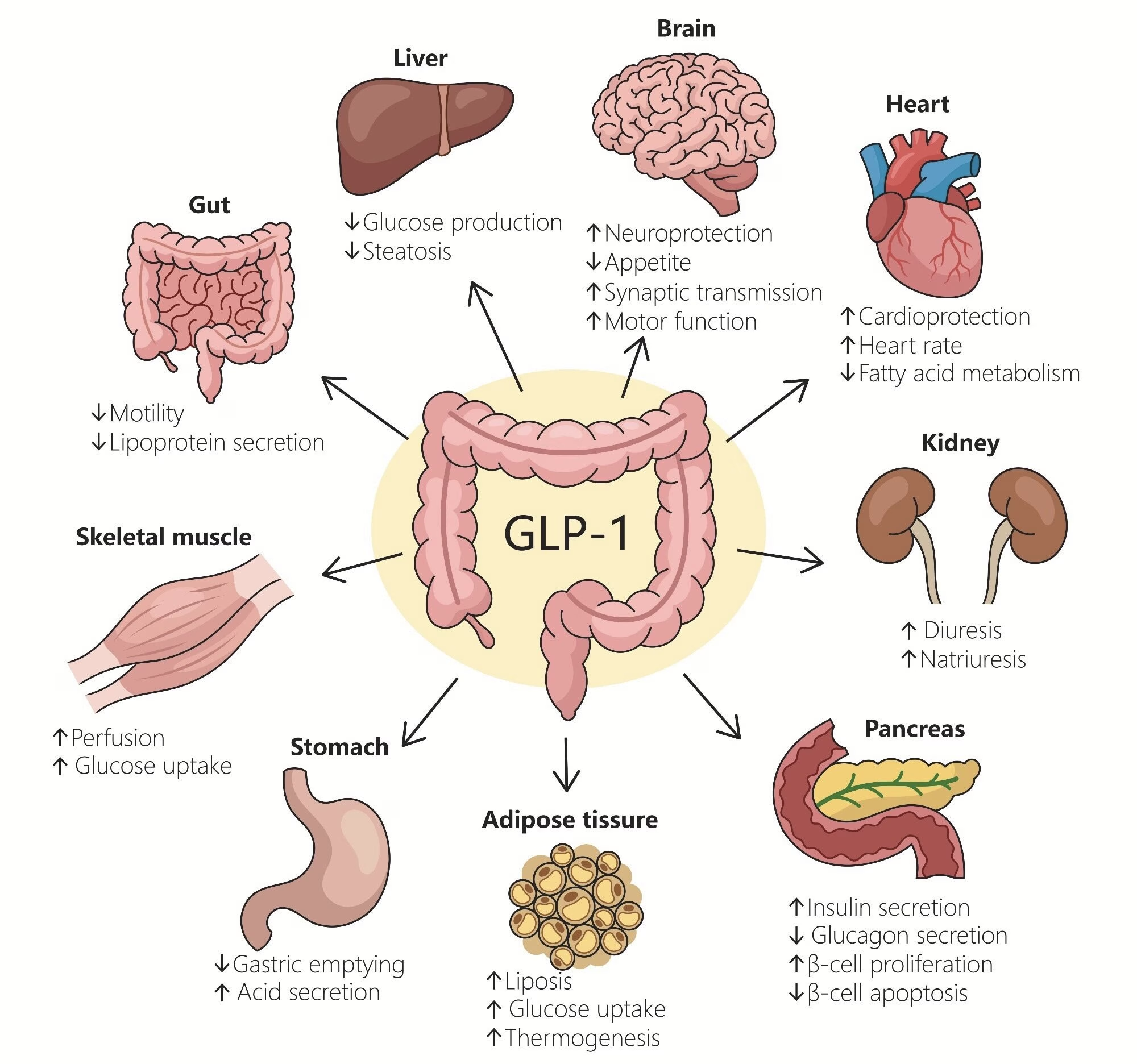
What is GLP-1 & how do these drugs work?
GLP-1 — glucagon-like peptide-1 — is a hormone your intestines produce naturally every time you eat. Its job is to help your body process the food you’ve just consumed and regulate blood sugar levels. The drugs work by mimicking this hormone at levels far above what your body would produce naturally.
There are three key mechanisms worth understanding:
1. Slowing gastric emptying
Food moves out of your stomach more slowly. You feel fuller for longer, and blood sugar rises more gradually after a meal. At the high doses now prescribed for weight loss, this effect is dramatic — food that would normally clear your stomach in three to four hours may sit there for much longer, which is also why persistent nausea is one of the most common side effects.
2. Suppressing glucagon
Glucagon is a hormone made in the pancreas that tells your liver to release stored glucose into the bloodstream. By suppressing glucagon, GLP-1 keeps blood glucose in check — and, critically, reduces insulin levels. Lower insulin means your body shifts toward burning fat rather than storing it.
This is actually a common point of confusion: many people assume these drugs lower blood sugar by stimulating more insulin. Research from the Astrup laboratory in Denmark — one of the world’s leading GLP-1 research groups — has shown the opposite. GLP-1 infusion in humans reduced both blood glucose and insulin levels, primarily by suppressing glucagon secretion and reducing gastric emptying. Less insulin, not more, is the metabolic benefit.
3. Reducing hunger signals in the brain
GLP-1 acts directly on the hypothalamus — the brain’s hunger and satiety centre — reducing appetite and increasing feelings of fullness. This works separately from the gut-slowing effect, so you get two appetite-suppression mechanisms working simultaneously. That’s a powerful combination, and it’s what drives the significant caloric reduction people experience on these drugs.
The thing most people get wrong about fat loss
Before we can properly evaluate any weight loss intervention — medication, surgery, or otherwise — we need to be precise about what we’re actually trying to achieve. And this is where a crucial piece of biology gets overlooked.
The goal is not to lose weight. The goal is to shrink fat cells. These are not the same thing.
Fat mass can increase in two completely different ways:
- Hypertrophy — existing fat cells get larger. Each cell stores more fat and swells.
- Hyperplasia — new fat cells are formed, so the total number increases.
In most people with metabolic disease or obesity, the primary mechanism is hypertrophy: a relatively fixed number of fat cells that have become excessively enlarged.
Here’s why this matters so much: it is the size of individual fat cells — not the total number or even the total fat mass — that drives metabolic dysfunction. When fat cells become enlarged, they become insulin-resistant. They release pro-inflammatory signals into your bloodstream. They stop safely storing energy and start causing harm. You can have a high total fat mass with small, healthy fat cells and be metabolically sound. You can have a relatively normal weight but large, dysfunctional fat cells and be in serious metabolic trouble.
The therapeutic goal, therefore, is to shrink fat cells. Not remove them, not freeze them off, not cut them out. Shrink them.
Why liposuction fails metabolically
This principle is illustrated perfectly by liposuction. Liposuction removes subcutaneous fat — the fat stored under your skin on the abdomen, thighs, and buttocks. But that’s actually the metabolically safer fat depot. The problematic fat — visceral fat, the fat wrapped around your internal organs — is deep inside the abdominal cavity and completely untouchable by liposuction.
When you remove fat cells from the wrong place, the body still wants to store the same amount of energy — because nothing about your diet or hormones has changed. The remaining fat cells compensate by increasing in size. Research confirms this: people who have liposuction show no improvement in cardiometabolic markers. They lost fat the wrong way.
The same fundamental limitation applies to the weight that returns after bariatric surgery, and to caloric restriction without hormonal support: unless the underlying drivers of fat cell enlargement are addressed, the body finds a way back.
The metabolic framework that actually works
So if shrinking fat cells is the goal, how do we achieve it? Here is the framework I work with clients, grounded in well-established biochemistry.
Fat storage is an insulin-dependent process
Without insulin, the fat cell cannot import and store energy — regardless of how many calories you consume. Insulin is the gatekeeper of the fat cell. And the primary driver of insulin secretion is dietary carbohydrate — specifically refined, processed, rapidly absorbed carbohydrates.
This is why the conventional approach of starting with caloric restriction so consistently fails over the long term. If you cut calories but your insulin remains elevated, your body is still directing energy toward fat storage rather than circulation. Your brain detects the low energy available and does exactly what it’s designed to do: it drives hunger. Harvard researcher Dr David Ludwig’s group has demonstrated this directly — measuring actual blood energy availability across different dietary conditions and finding that high-insulin states reduce available energy even when caloric intake is unchanged.
Hunger always wins against restriction if the hormonal environment hasn’t changed. This is not a willpower problem. It’s biology.
Lower insulin first. Then let caloric reduction follow naturally.
The two-step path to fat cell shrinkage
Step One is lowering insulin — primarily by reducing refined carbohydrate intake. When insulin falls, several things happen simultaneously:
Metabolic rate increases, sometimes meaningfully above baseline.
- Fat cells begin releasing stored energy into circulation rather than hoarding it.
- The liver begins producing ketone bodies as a by-product of accelerated fat burning, and a proportion of those ketones are excreted — calories literally lost from the system.
- The brain detects abundant circulating energy and reduces hunger drive. Satiety improves without deliberate restriction.
The practical prescription for Step One: control carbohydrates, prioritise protein, and don’t fear dietary fat. Avoid the carbs that come in bags and boxes with barcodes. Eat whole fruits and vegetables rather than drinking them. Protein and fat have minimal effects on insulin and are the macronutrients that support satiety, lean mass, and hormonal function.
Step Two — if needed — is reducing caloric intake through structured intermittent fasting, applied after insulin is already managed. In this hormonal environment, the body burns fat efficiently, hunger is genuinely attenuated, and lean mass is far better preserved.
What about exercise?
Exercise is essential — for cardiovascular health, strength, insulin sensitivity, and lean mass preservation. But it is not, in itself, an effective fat-loss strategy. The American College of Sports Medicine has acknowledged this. You cannot reliably out-exercise a high-carbohydrate, high-insulin diet. The mantra I work by: exercise to be healthy and strong; eat strategically to be lean. These are different goals requiring different tools.
The risks most prescribers aren’t discussing
Every drug involves a trade-off. Here is what I believe every person considering GLP-1 receptor agonists deserves to know before they start.
1. Mental health: depression, anxiety, and anhedonia
A large cohort study comparing patients on GLP-1 receptor agonists with those not taking them found a 195% increased risk of major depression, a 108% increased risk of anxiety disorders, and a 106% increased risk of suicidal behaviour. These are not marginal findings. They’re clinically significant.
The likely mechanism is anhedonia — a reduced ability to experience pleasure across the board. These drugs broadly suppress the brain’s reward circuitry, not just selectively for food. People may find that activities they used to love — social connection, hobbies, physical activity, intimacy — simply feel less compelling. That flattening of experience is almost certainly contributing to the elevated rates of depression.
This risk appears substantially higher in people with any pre-existing history of mood disorders — a group that is often excluded from clinical trials but very much present in real-world prescribing.
2. Loss of libido and sexual dysfunction
Less commonly discussed but worth knowing: loss of libido and sexual dysfunction are documented side effects. When the brain’s reward and pleasure pathways are broadly suppressed, sexual desire and function are affected. This matters for quality of life and, practically, for whether people stay on the medication.
3. Muscle and lean mass loss
Studies consistently show that approximately 40% of the total weight lost on semaglutide comes not from fat, but from fat-free mass — muscle, bone, and other lean tissue. This is largely a consequence of profound caloric deficit rather than a direct effect of the drug (similar proportions are seen with bariatric surgery and severe caloric restriction). But that context doesn’t make it less serious.
Muscle mass is one of the strongest independent predictors of longevity. Losing it worsens insulin sensitivity, reduces your resting metabolic rate, and makes long-term weight management harder. And critically: when the drug is eventually stopped, fat mass returns rapidly. Muscle and bone — especially in people past middle age — may not.
4. New fat cell formation
Liraglutide has been shown to stimulate the formation of new fat cells by activating the PPARγ receptor. While you’re on the drug, those new cells stay small because insulin is suppressed and intake is reduced. But when the drug is stopped, and old eating patterns return, those extra fat cells fill. Combined with the lean mass lost during treatment, you can end up with a body composition that’s worse than where you started: more fat cells, less muscle, lower metabolic rate.
5. The drugs stop working
A two-year clinical trial found that cravings suppression and hunger reduction — the primary benefits — had largely returned to baseline by the two-year mark. This is a fundamental principle of receptor biology: sustained overstimulation leads to reduced receptor sensitivity over time. These drugs are no exception.
6. Most people stop within two years anyway
Perhaps the most sobering statistic: over 70% of patients in the US and UK stop these medications within two years. Sometimes, because of cost or access. Sometimes, because of persistent nausea. Sometimes, because of mood-related effects. Sometimes, simply because the drug has stopped working as well.
When they stop, the fat returns rapidly — but the lean mass does not come back nearly as easily. That asymmetry is the real long-term problem.
The craving research that changes how we should think about these drugs
In 1996, researchers published a study in the journal Gut that I think is genuinely underappreciated. They measured GLP-1 responses to carbohydrate-rich meals in lean versus obese individuals. What they found was striking:
- Lean participants showed a robust GLP-1 response after eating — satiety was signalled, hunger was suppressed, and they naturally stopped eating.
- Obese participants eating the same meal showed a blunted or absent GLP-1 response — the stop signal never arrived. Appetite continued. Cravings persisted.
Professor Arne Astrup — one of the world’s leading GLP-1 researchers — has framed this compellingly: what if obesity, for many people, is not primarily a failure of willpower, but a failure of adequate GLP-1 signalling in response to carbohydrate?
That reframe changes the entire conversation about these medications.
And it connects to another well-established piece of neuroscience: humans don’t crave fat. They don’t crave protein. The consistent finding across craving research is that we crave carbohydrates—refined, processed, and highly palatable. The salty-crunchy variety or the sweet-dense variety. GLP-1 receptor agonists, by acting on the hypothalamus, directly attenuate those cravings. That may actually be their most clinically valuable property — if we use it correctly.
A smarter way to use these medications
Here is what a better use of GLP-1 medications looks like — and there are clinicians doing exactly this right now, with results that outperform standard protocols.
Instead of prescribing full therapeutic doses as an indefinite weight-loss drug, what if we used a much lower dose — a micro-dose — for a defined and limited period, with a very specific purpose: to help the patient break their carbohydrate addiction and transition to a genuinely lower-carbohydrate diet?
Why micro-dosing makes sense
A microdose is sufficient to attenuate carbohydrate cravings without producing the full gastric slowing, nausea, anhedonia, libido suppression, and lean mass loss that come with high-dose therapy. The mechanism you’re targeting is hypothalamic craving suppression, not gastric paralysis.
A 90-day protocol gives enough time for new dietary habits to become genuinely established — the neuroscience of habit formation supports that window. At the end of the cycle, you wean off the drug. If habits have taken hold, the patient continues without pharmacological support. If cravings begin to return, another short cycle is an option.
The drug becomes a tool with an exit strategy — not a lifelong dependency.
Early clinical data comparing low-dose semaglutide combined with a structured low-carbohydrate programme to standard-dose semaglutide alone show results that outperform the standard protocol — with better patient experience, fewer side effects, and stronger long-term dietary adherence.
Why the dietary approach matters more than the drug
The dietary strategy that must accompany this is low-carbohydrate eating — not caloric restriction, not low-fat. A real reduction in refined carbohydrates, with adequate protein, adequate fat, and whole food sources. This is what lowers insulin sustainably. This is what creates the metabolic environment in which fat cells genuinely shrink.
And importantly, a low-carbohydrate approach substantially preserves lean mass — something high-dose GLP-1 therapy on a conventional diet consistently fails to do. Research from BYU has shown that beta-hydroxybutyrate — the primary ketone body produced during low-carb eating — directly protects muscle cells from breakdown. Preserving muscle mass is not a cosmetic concern; it is central to long-term metabolic health, insulin sensitivity, and longevity.
What to do with all of this
Whether you’re currently on GLP-1 medications, considering them, or advising someone who is, here are the questions worth asking:
- What is the dietary plan that accompanies the prescription? ‘Eat less’ is not sufficient — it doesn’t address the hormonal environment that drove fat storage in the first place.
- Is there a timeline and an exit strategy, or is this an indefinite prescription?
- Is anyone monitoring muscle mass, mood, and libido? If not, those conversations need to happen.
- Has anyone discussed a structured low-carbohydrate approach as either an alternative or a complement?
GLP-1 medications can be a powerful short-term tool. But no drug can replace the metabolic foundations of genuine health.
The goal — always — is to shrink fat cells by lowering insulin, through real food, with lean mass preserved. Medications can support that journey. They cannot substitute for it.
Want to go deeper?
I have also explored this topic in a recent article published in Brainz Magazine. You can read it here:
→ 7 Things You Need to Know About Ozempic, Wegovy, and the Weight Loss Revolution